AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Magnesium atomic structure8/31/2023 
From both the electronic configurations it is clear that magnesium has two valence electrons whereas oxygen has six valence electrons. On the other hand, the atomic number of oxygen is 8 by which its electronic configuration is 1s2 2s2 2p4. Since sodium has 11 protons, the number of neutrons must be 23 – 11 = 12 neutrons. The atomic number of magnesium is 12 where its electronic configuration is 1s2 2s2 2p6 3s2. 
The mass number of an element tells us the number of protons AND neutrons in an atom (the two particles that have a measurable mass). This tells us that sodium has 11 protons and because it is neutral it has 11 electrons. 12 electrons (white) occupy available electron shells (rings). The nucleus consists of 12 protons (red) and 12 neutrons (orange). We know that the atomic number of sodium is 11. Diagram of the nuclear composition, electron configuration, chemical data, and valence orbitals of an atom of magnesium-24 (atomic number: 12), the most common isotope of this element. The mass number is given at the top left of the elements symbol, for example, sodium has a mass number of 23. This is why elements in the same group of the Periodic Table have similar properties. It is the number of outer electrons that give an element its chemical properties. We’ll use a Bohr diagram to visually represent where the electro. The second energy level is also full, holding eight electrons and one electron remaining in the outer energy level. In this video well look at the atomic structure and Bohr model for the Magnesium atom (Mg). The first two fill the innermost energy level. The first energy level (the one nearest the nucleus) can hold a maximum of two electrons with the others being able to hold up to a maximum of 8 electrons (only true for the first 20 elements).Ī sodium atom contains 11 electrons orbiting a nucleus in 3 levels, 2 in the lowest, then 8, then 1 These energy levels can only hold a certain number of electrons. All the electrons are arranged into energy levels. It is a shiny gray metal having a low density, low melting point and high chemical. A possible crystal structure of Magnesium is hexagonal close-packed structure. 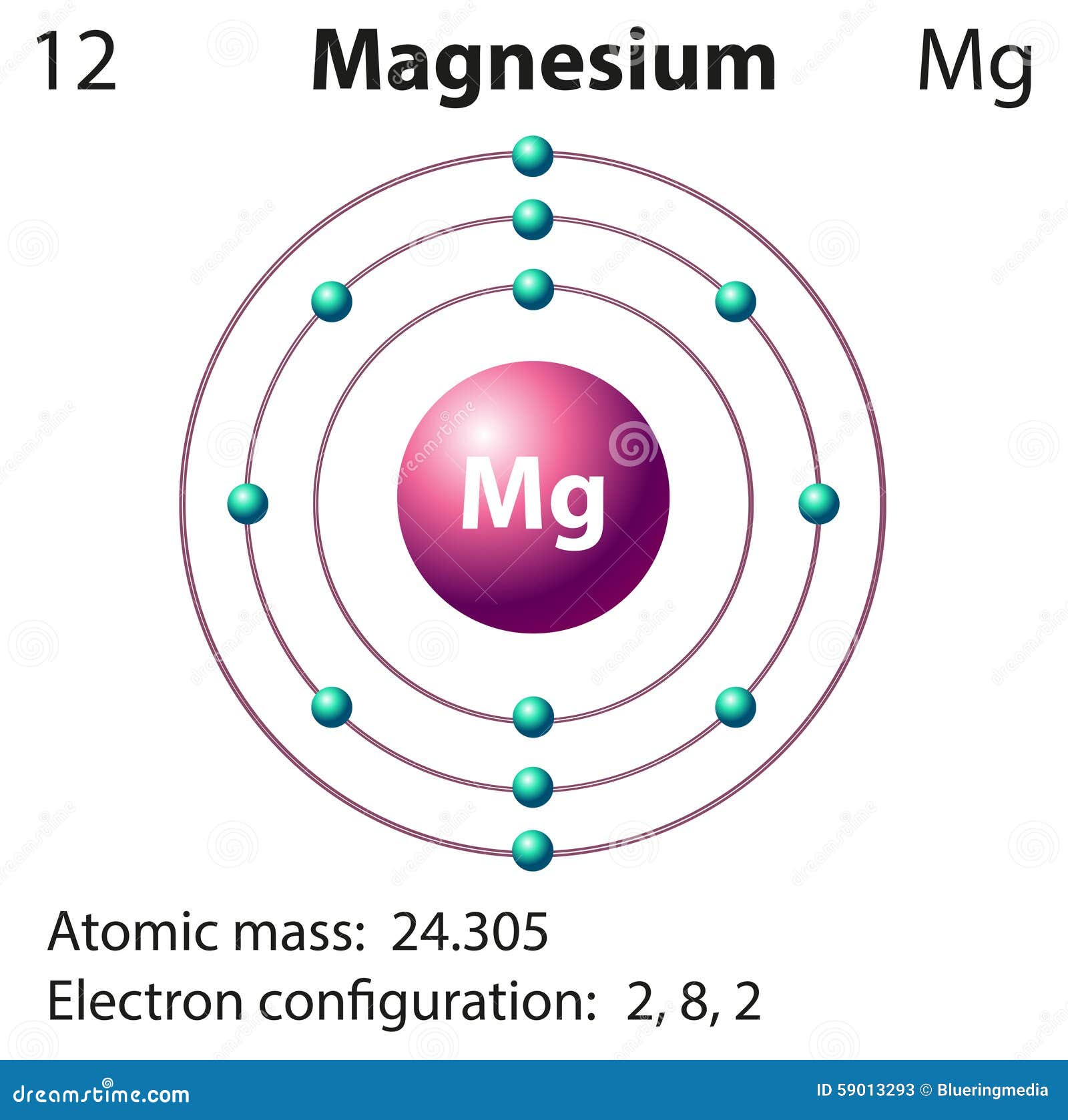
The electron arrangement of all atoms can be found in the data booklet. Atomic Data of Magnesium (Element 12) Atomic structure Number of Electrons Number of Electrons, 12 Number of Neutrons, 12 Number of Protons, 12. Magnesium is a chemical element with the symbol Mg and atomic number 12. Since atoms are neutral, we know then that sodium atoms must also have 11 negative electrons to cancel the charge from 11 positive protons. This is written at the bottom left hand side of the symbol. The atomic number of an element tells you how many protons that the element has.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
